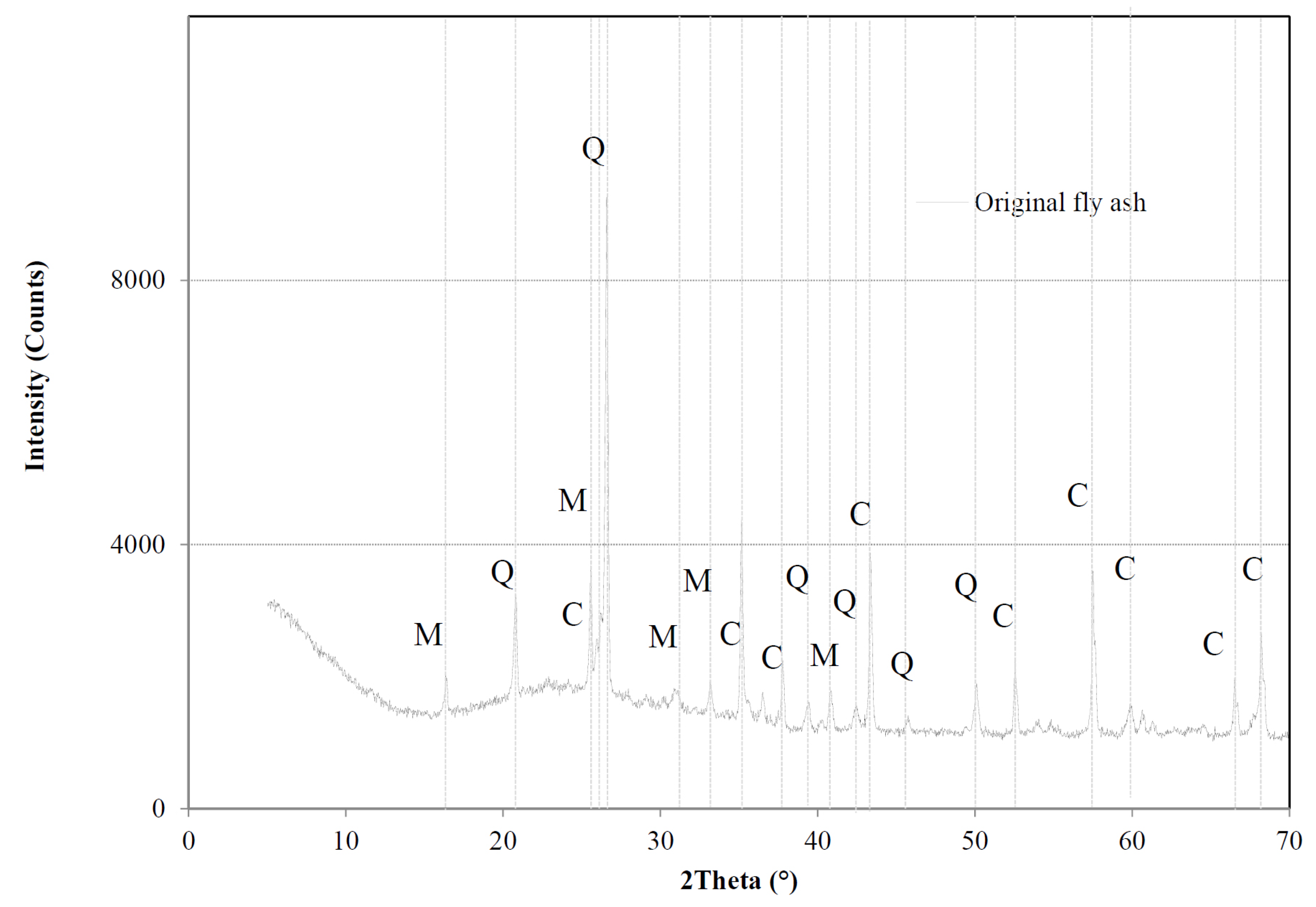
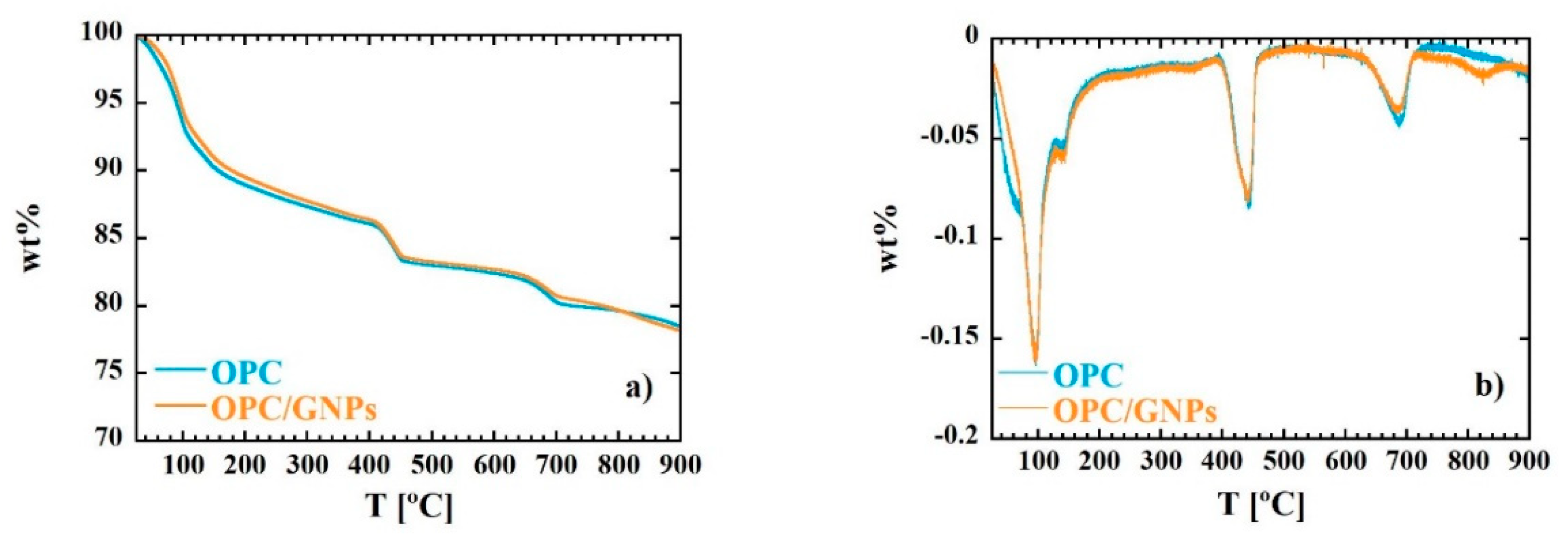
The main objective of the thesis project was to develop a sustainable and extrapolable method on an industrial scale for the synthesis of hydrated calcium silicates, ie, xonotlite and tobermorite. The main advantage of this methodology is linked to the ultra-fast reaction kinetics, which allows the synthesis of these mineral phases in supercritical water in just a few seconds. Also, in addition to very short synthesis times, synthetic minerals are characterized by high crystallinity and purity, unlike their natural counterparts which are often found mixed with other phases or impurities. Thanks to this property, synthetic xonotlite and tobermorite can be used to deepen the structure of hydrated calcium silicates, which is not yet fully elucidated. Subsequently, highly crystalline xonotlite and tobermorite have been used in two different fields of application. The first is linked to the use of mineral phases in the form of seeds to accelerate the hydration of the cement in order to develop a denser and more resistant cement matrix. The second application considers the use of xonotlite as a precursor to produce, following its dehydration, another calcium silicate, wollastonite. Finally, with the aim of expanding the production of synthetic minerals to industrial requirements, the environmental impact of this methodology has been studied through life cycle analysis. The impacts of the most relevant parameters, such as energy, nature and concentration of precursors, were analyzed to allow possible optimization.
VALENTINA MUSUMECCI
PhD student (U. Bordeaux-UPV/EHU) Supercritical Fluids. Nano-tobermorite, nanoxonotlite, nanoCSH